A Compound That Contains Only Carbon Hydrogen and Oxygen
The energy for this comes from. In the Light Independent Process carbon dioxide from the atmosphere or water for aquaticmarine organisms is captured and modified by the addition of Hydrogen to form carbohydrates general formula of carbohydrates is CH 2 O n.

Hydrocarbons Class 11 Notes Chemistry Chapter 13 Learn Cbse Chemistry Notes Chemistry Chemistry Basics
An electric current splits water into hydrogen and oxygen.

. Some compounds also contain ide for instance OH hydroxide. Other chemical substances commonly encountered in pure form are diamond carbon gold table salt sodium chloride and refined sugar sucrose. With the exception of the lighter inert gases helium He neon Ne argon Ar and krypton Kr oxygen O forms at least one binary oxide with each of the elements.
Other elements such as nitrogen about 05 sulfur 05 oxygen 1 and metals such as iron nickel and copper less than 01 can also be mixed in with the hydrocarbons in small amounts. A molecule may be homonuclear that is it consists of atoms of one chemical element as with two atoms in the oxygen molecule O 2. Crude oil is composed of hydrocarbons which are mainly hydrogen about 13 by weight and carbon about 85.
Water which is a chemical compound of hydrogen and oxygen in the ratio two hydrogen atoms for every oxygen atom contains H 2 O molecules. The resulting synthesis gas contains hydrogen and carbon monoxide which is reacted with steam to separate the hydrogen. A molecule is an electrically neutral group of two or more atoms held together by chemical bonds.
The incorporation of carbon dioxide into organic compounds is known as carbon fixation. Ate is employed when there are more oxygen atoms present in a compound and ite is used when number of oxygen atoms present in a compound is less. Although the formula for sodium chloride is NaCl the compound does not contain actual NaCl molecules.
Sodium chloride is a chemical compound formed from sodium Na and chlorine Cl in a 11 ratio. If the electricity is produced by renewable sources such as solar or wind the resulting hydrogen will be considered renewable as well and has numerous emissions benefits. Scheele in 1771 by decomposing fluor-spar with concentrated sulphuric acid a method still used for the commercial preparation of the aqueous solution of the acid the mixture being distilled from leaden retorts and the acid stored in leaden or gutta.
The air that comes out of your mouth when you exhale contains carbon dioxide and carbon dioxides chemical formula is CO 2 because there is one carbon atom bonded to two oxygen atoms. Oxide any of a large and important class of chemical compounds in which oxygen is combined with another element. Both metals and nonmetals can attain their highest oxidation states ie donate their maximum.
A common example of a chemical substance is pure water. C To name binary compounds. Or it may be heteronuclear a chemical compound composed of more than one element as with water two hydrogen atoms and one oxygen.
Only one compound of hydrogen and fluorine is known namely hydrofluoric acid HF or H 2 F 2 which was first obtained by C. It always has the same properties and the same ratio of hydrogen to oxygen whether it is isolated from a river or made in a laboratory. B If the compound contains polyatomic ion then the last three alphabets of a non-metal are replaced with ate or ite.
Rather it contains equal. Simple or seemingly pure substances.

Organic Chemistry Periodic Table Organic Chemistry Chemistry Chemistry Help

Environmental Chemistry Cbse Notes For Class 11 Chemistry Learn Cbse 11thchemistrynotes Enviro Environmental Chemistry Chemistry Class Chemistry Class 11

Pin On Reactions Of Carboxylic Acids And Their Derivatives Practice Problems

Pin On Reactions Of Carboxylic Acids And Their Derivatives Practice Problems

Elements Compounds And Mixture Elements Compounds And Mixtures Compounds And Mixtures Study Notes

Pin On Acids Bases And Solutions

Pin By Natalie Chalmers On Chemistry Science Notes Math Books Science Questions

Venn Diagram Of Element And Compound Differences And Similarities Venn Diagram Chemistry Basics Diagram

Dalton S Atomic Theory Reading Comprehension Practice Science Teacher Resources Reading Passages

Chemical Bonding And Molecular Structure Cbse Notes For Class 11 Chemistry 8 11th Chemistry Chemistry Chemistry Notes

Electrolysis Aqueous Solutions 4 Science Chemistry Solutions Chemistry
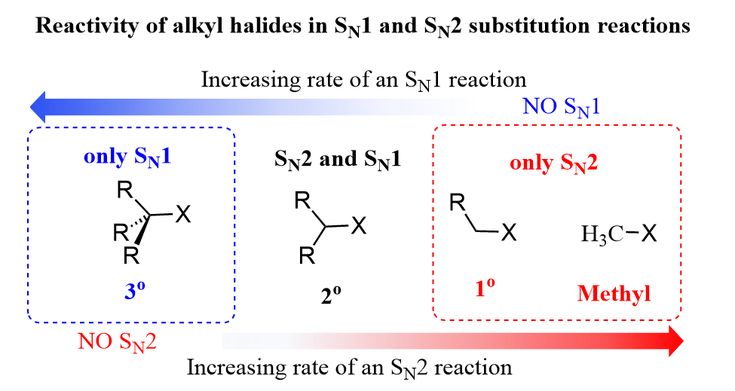
Sn1 Sn2 Reactivity Of Alkyl Halides Organic Chemistry Organic Chemistry Study Organic Chemistry Mechanisms

Welcome To Learnapchemistry Com Chemistry Classroom Teaching Chemistry Worksheet Template

Pin On Acids Bases And Solutions

Naming Isomers 3 Structural Formula Chemistry Study Tips

Highhill Homeschool Cell Unit Study Week 2 Atoms Study Unit Science Projects Science Projects For Kids

Atomic Orbital Easy Science Electron Configuration Easy Science Atom

Hydrocarbon Easy Science Biology Notes General Knowledge Book Easy Science

Comments
Post a Comment